Chemotropism – response to pheromone gradients
Cells interact with their environment. They are able to perceive neighbors and respond to external signals that guide their behavior. More often than not, the perceived signals are directional and lead to a cellular response whose polarity is physiologically critical. Examples of such chemotropic behaviors include those of leukocytes migrating towards and responding to antigens, amoebae responding to a cue for aggregation, neurons extending growth cones to form neural networks, pollen tubes growing in the stigma towards the egg cell or fungal hyphae creating connected mycelia. The meeting of gametes during sexual reproduction is a particularly important form of chemotropic behavior, which culminates in their fusion to form the zygote.
Fungal cells exhibit a chemotropic response to pheromone gradients expressed by partner cells. During sexual differentiation, cells of distinct mating types (P and M) secrete pheromones recognized by cognate G-protein coupled receptors on the partner cell. This activates a common signaling pathway that involves activation of Ras GTPase and MAP kinase cascade. We are exploring the mechanisms by which cells detect pheromone gradients and re-orient their polarization machinery towards a mating partner.
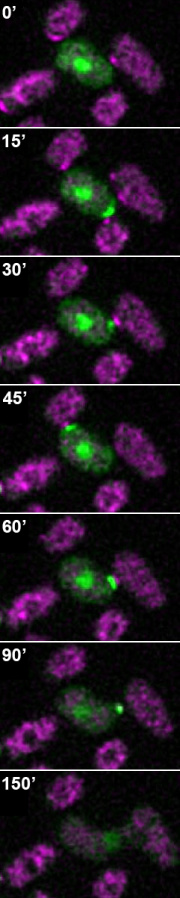
We have shown that, in the early stages of the mating process before cells have selected a mating partner, or upon exposure to low-level pheromone levels, fission yeast cells display a dynamic site of polarization. The small GTPase Cdc42, a central eukaryotic regulator of cell polarization, forms a cortical patch that appears and disappears dynamically at the plasma membrane. Ras GTPase, which promotes Cdc42 activity, is also active at these patches. The timelapse on the right shows cells of distinct mating types, in which activators of Cdc42, which are co-dynamic with Cdc42, have been labelled green or red. The green-labeled cell appear to explore several potential partner before settling on one for fusion.
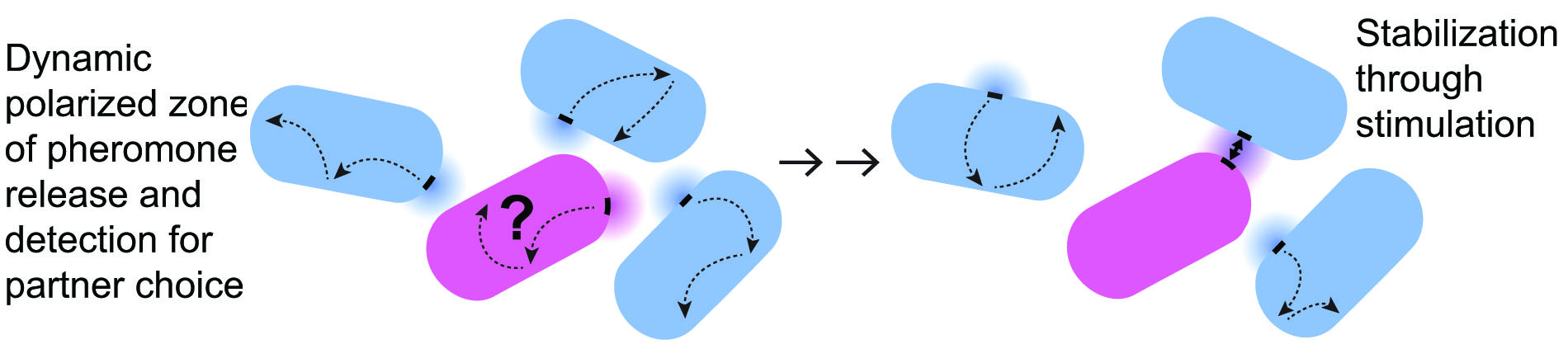
These patches are fully developed polarity sites, from which pheromones are locally secreted. Components of pheromone sensing are also present at the patches, which become more static when perceiving higher pheromone concentrations. Thus, each patch is a mobile communication site, where pheromone signals are sent and locally interpreted. We are exploring the model that yeast cells form pairs through a “speed-dating” strategy. The cortical communication patch is mobile, but stimulation by pheromone released by a partner zone stabilizes it. This leads to positive feedback stimulation, where the encounter of patches from two partner cells stabilizes them through local pheromone release. Such feedback is also critical in the transition from growth to cell-cell fusion.